Browse through our Journals...
COMPARISON OF VISUAL ESTIMATION OF BLOOD LOSS WITH SERIAL HEMOGLOBIN AND HEMATOCRIT ESTIMATION IN SUPRATENTORIAL CRANIOTOMY
Abbasi S, FCPS (Anaesthesiology)
Assistant Professor
Department of Anaesthesia
Aga Khan University
Khan FA, FRCA, FCPS
Professor
Department of Anaesthesia
Aga Khan University
Adil S. FCPS
Associate Professor
Department of Hematology and Oncology
Aga Khan University
Enam SA. MD, Phd, FRCS(I), FRCS (Can), FACS ;
Associate Professor
Department of Neurosurgery
Aga Khan University
Corresponding Author:
Dr. Abbasi S
Department of Anaesthesia
Aga Khan University
Stadium Road, P.O Box 3500 Karachi 74800, Pakistan
ABSTRACT:
Estimate of actual blood loss during neurosurgery with traditional methods like measuring the loss of blood in suction bottles, drapes and swabs is difficult. The accurate assessment of the blood loss is of critical importance in these patients in order to maintain oxygen delivery to the brain. Our aim was to find out a relationship between hemoglobin/ hematocrit values based on visual estimation of blood loss versus laboratory calculation during supratentorial craniotomies.
Fifty six elective and emergency patients, above 18 years of age, undergoing supratentorial craniotomy were recruited. In addition to the demographic information, baseline hemoglobin, hematocrit, allowable blood, visual estimated blood loss, estimated hemoglobin, calculated Hemoglobin / hematocrit and blood transfusion were documented on hourly basis intra-operatively.
54/56 cases were included, out of which 57% patients were male. Blood sampling was done on 178 occasions in these 54 cases. In 24% readings estimation of hemoglobin fell within the accurate range whereas 62% overestimation of hemoglobin and 14 % underestimation of hemoglobin was observed. The mean (SD) for calculated blood loss was 943.5 (956) mls and the mean for estimated blood loss (SD) was 681 (565) mls and the difference between the two was significant (p value 0.013). In 3 cases we intervened, as Hb was less than 8.0gm/dl.
The estimated blood loss is not a good predictor of calculated blood loss. Laboratory investigation was found to have significant difference with the routine method of visual estimation.
Key Words: Blood loss, craniotomy, hemoglobin
INTRODUCTION:
Accurate assessment of blood loss is a problem in specialties like neurosurgery, head and neck surgery and orthopaedics where large volumes of fluid is used for irrigating wounds during surgery. It is also a problem in cases where blood is mixed with body fluids e.g. in obstetrics and in intracranial and spinal procedures. Inaccuracies in blood loss estimation eventually lead to inaccurate decisions in transfusing patients. Inappropriate decision of transfusion may lead to low hemoglobin values with the potential of tissue injury (1). The actual level of hemoglobin at which tissue injury starts to threaten tissue oxygenation is still uncertain.
The assessment of blood loss is of critical importance in intra cranial procedures in order to maintain oxygen delivery to the brain. This acts as a guide to both timely management and further arrangement of cross matched blood during neurosurgery. The current literature in neuroanaesthesia is deficient in accurate assessment of intraoperative blood loss.
In this study we have attempted to find any relationship between hemoglobin (Hb)/ hematocrit (Hct) values based on visual estimation of the blood loss by the anaesthetist and Hgb/ Hct measurements based on actual Hb/ Hct sampling at the same point in time.
METHODS:
The study was conducted at a university teaching hospital where four elective lists are dedicated to neurosurgery every week. It was designed as a double blind, prospective, non randomized, controlled comparative study in which each patient acted as his or her own control. The study was approved by the ethical committee of the institution and written informed consent was obtained from all patients. If the patient was unable to give consent due to depressed conscious status, consent was taken from the legal guardian. Consent was also taken from the participating consultant anaesthetists.
Sample size calculation was based on the assumption that the anticipated correlation co-efficient between readings of two methods is 0.5 at a power of 80 % and level of significance of 5 %. Fifty two cases were required but we recruited 56 patients to compensate for any dropouts.
All ASA (American society of Anesthesiologist) I, II, III and IV adult patients, age more than 18 years undergoing elective or emergency supratentorial craniotomy were included. Patients undergoing mini craniotomy or burr hole procedure or those with hypovolemia or hypervolemia (hematocrit values less than or more than three times the hemoglobin) and with history of active bleeding within last 48 hours were excluded. Convenient sampling strategy was used.
Two separate forms were designed for data collection. Primary anaesthetists were requested to fill the first form (Form I) which included patients medical record number, age, gender, ASA class, surgical procedure, baseline hemoglobin (Hb) and hematocrit (Hct) and allowable blood loss. Allowable blood loss was calculated at a Hct of 30% (2, 3) with the help of the following formula VL= EBV x Ho – HF/Ho in which VL= allowable blood volume, EBV= estimated blood volume, Ho= patient’s initial hematocrit (or hemoglobin concentration) and Hf = patient’s minimum allowable hematocrit (or hemoglobin concentration) (4).
Central venous pressure measurement is also used in cases for further accuracy of volume status.
Intra operatively the primary anaesthetist conducting the case listed the hourly visual blood loss estimation (EBL) over the preceding hour, the estimated Hb (EHb) and Hct based on the visual blood loss estimation and any blood transfusion administered. Blood soaked in drapes, gauzes, cotton balls and surgeon’s gown, collected in suction bottle and shed in the bucket were accounted for estimation of blood loss. A dedicated research assistant was responsible for sampling and stat result collection. Calculated blood loss (CBL) represented the blood loss which was calculated with the help of results of hemoglobin provided by laboratory using Coulter LH750. A drop of hemoglobin by 1gm/dl is taken as blood loss of 500 mls. Blinding was ensured by only the principle investigator (who was not directly involved in case management) and research assistant being aware of the laboratory results of Hb / Hct (Calculated Hb) which was noted on a separate form. The primary anaesthetist was informed if laboratory hemoglobin turned out to be less than 8 gm/dl for the safety of the patient.
Statistical Package of Social Sciences version 16.0 was used for data entry and statistical analysis. Mean, standard deviation (S.D,), range, percentile, percentage were used to analyze basic demographics. Paired t-test was used to compare the average difference between CBL and EBL
RESULTS:
Fifty six patients were included in the study. Mean age of patients was 41 years with standard deviation of 12.5 and range of 19 to 67 years. The 25th percentile was 32 years and 75th percentile was 51 years. Thirty two (57%) patients were male and 24 (43%) were female. All these patients belonged to ASA class II (n=30), III (n=24) and IV (n=2) (table 1).
Two cases where surgery was terminated in less than one hour were excluded from statistical analysis in order to prevent any outlier effect.
The duration of surgery in 54 cases varied between 2 to 6 hours with a mean of three hrs (SD of 1 hr). Blood sampling was done on 178 occasions in these 54 cases. Difference of estimated and calculated hemoglobin was calculated in these readings. These differences were arbitrarily divided into different ranges. Difference of -0.5 to +0.5 g/dl in estimated and calculated hemoglobin was considered nearly accurate, difference of hemoglobin +0.6 to +1.0 g/dl, +1.1 to +1.5 g/dl, >+1.5 g/dl considered overestimation and difference of hemoglobin –0.6 to -1.0 g/dl, -1.1 to -1.5 g/dl and < - 0.5 g/dl as underestimation. This information is presented in Fig 1. In twenty four percent readings estimation of hemoglobin fell within the accurate range, overestimation of hemoglobin found on 62% occasions and underestimation in 14% of the readings.
The distribution of estimated Hb and calculated Hb level by hour of operation showed a significant difference in estimated Hb and calculated Hb in initial two hours of surgery (Fig 2). The difference reduced in the third and fourth hour.
The mean (SD) for CBL was 943.5 (956) mls and the mean for EBL (SD) was 681.3 (565) mls and the difference between the two was significant (p value 0.013). In 36 cases the anaesthetist mean estimated blood loss was less and in 18 cases it was above the CBL.
We found poor correlation with regression co efficient of 0.38 when estimated blood loss was compared with calculated blood loss through linear regression (Fig 3).
Hb dropped less than 8 gm/dl in only three patients in first, second and third hour of craniotomy. In these three cases study was terminated and the results were provided to the primary anaesthetist according to the protocol.
DISCUSSION:
Different methods are available for estimation of intra operative blood loss, but gravimetric measurement, hemoglobin calorimetry, electrical conductivity and osmolality have all failed to gain wide acceptance due to their limitations (5-9). In addition there is unavailability of specific equipment and personnel within operating rooms to perform these tests. Duration of surgery, cost constraints and requirement of continuous hemodynamic monitoring are other limitations. At present there is no gold standard to assess intra operative bleeding.
Accurate and timely estimation of intra operative blood loss is important for appropriate replacement with crystalloid or colloids, as well as early request for the release of packed RBCs or ordering further packed cells. Underestimation and inadequate replacement will primarily lead to hypotension and tachycardia. Secondarily very low hemoglobin possesses risk of tissue injury due to impaired oxygenation. Hare et al showed that there was a possibility of worsening brain injury at hemoglobin level well above the commonly accepted level of 7.0g/dl (10). Over estimation and over transfusion on the other hand will increase the myocardial workload due to hypervolemia and increased viscosity. Over estimation of blood loss will expose the patient to the risks of blood transfusion (11). It has been suggested that risks of transfusion increases with intra operative transfusion (12, 13).
Neurosurgical anaesthesia is associated with additional concerns related to blood loss estimation and transfusion. This includes inaccurate visual estimation of lost blood, inability to control bleeding by applying pressure and inability to judge sign and symptoms of hypoxia within the brain in an anaesthetized individual. Visual estimation of blood loss during neurosurgery should include blood in suction bottles diluted with irrigation fluid and cerebrospinal fluid, blood shed on floor, soaked surgical gowns, drapes and sponges. An additional problem in neurosurgery is dark operating rooms due to the use of microscopes.
Some work has been done on methods of blood loss estimation during different surgical specialties like orthopedic, urology and obstetrics (14, 15, 16). Some of these studies are clinical (17) whereas others have compared visual versus other methods of estimation for inter observer variability on objective structured clinical examination (OSCE) stations (18, 19). Clinical literature shows discrepancy between laboratory determination and visual estimation of blood loss during normal delivery (20). In our literature search we were unable to locate any study comparing visual and laboratory estimation of blood loss during neurosurgery.
In this study we found a significant estimation error of 76% (135/178) of over or under estimation of blood loss. On comparison there was poor correlation between EBL and CBL with regression co efficient of 0.38. This is similar to the results of Brecher and colleagues in urological surgery. They showed that CBL was on average 2.1 times the EBL during radical prostatectomy. They recommended that estimation of blood loss by mathematical models provided rapid estimation of blood loss (15).
We also observed that comparison of estimated and calculated hemoglobin showed a statistically significant difference of EBL and CBL during the first hour of surgery and this difference decreased over time and became negligible in the fourth hour of surgery. This can be explained by the fact that few anaesthetists practiced the concept of allowing loss of diluted blood initially followed by replacement with packed RBCs once haemostasis was secured.
There is one limitation to our study which was unavoidable. These estimations were done by four anaesthetist involved in neurosurgical anaesthesia, we therefore cannot eliminate inter observer variability and bias.
Further work needs to be done on this topic and future guidelines are required for intra operative hemoglobin measurement during supratentorial craniotomy. Visual estimation is an easy, cost free, acquirable and mostly practiced method of blood loss estimation but it still needs to be proven that laboratory estimation is a more accurate method for blood loss estimation during neurosurgery for maintaining better homeostasis and oxygenation in vital organs. Initial work needs to focus on whether a difference exists between the two methods. If yes, than which of these methods can be relied upon. Our study focused on the first question and we concluded that on 62% occasions there were underestimation of blood loss and in 14% overestimation of blood loss compared to laboratory measurements of hemoglobin and hematocrit.
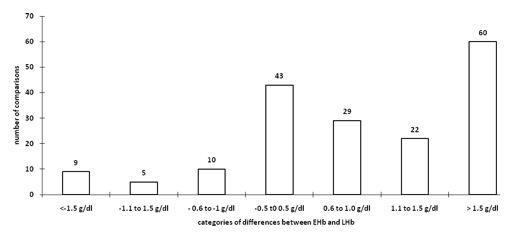
Figure 1: frequency of difference in estimated (EHb) and laboratory hemoglobin (LHb) on 178 ocassion
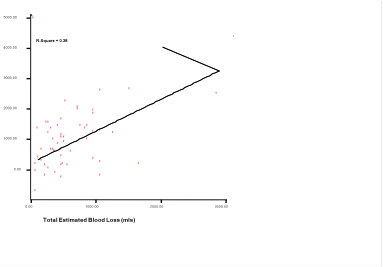
Figure 2: Comparison of calculated and estimated blood loss (EBL and CBL) by linear regression graph. The line of identity (-) is placed where EBL matches with CBL.
ACKNOWLEDGEMENT:
The study was funded by the Seed money Grant provided by Aga Khan University. We are very thankful for the statistical help provided by Mr. Syed Iqbal Azam, Assistant Professor Department of Community Health Sciences Aga Khan University.
Legends for figures:
- Frequency of difference in estimated (EHb) and laboratory hemoglobin (LHb) on 178 ocassion
- Estimated and Laboratory Hemoglobin according to hours of operation.
- Comparison of calculated and estimated blood loss (EBL and CBL) by linear regression graph. The line of identity (-) is placed where EBL matches with CBL.
REFERENCES:
- Beattie WS, Karkouti K, Wijeysundera DN, Tait G. Risk associated with preoperative anemia in noncardiac surgery: a single-center cohort study. Anesthesiology 2009; 110:674-581.
- The Clinical Use of Blood in Medicine, Obstetrics, Paediatrics, Surgery & Anaesthesia, Trauma & Burns. Geneva: World Health Organization; 2001.
- Razvi K, Chua S, Arulkumaran S, Ratnam SS. A comparison between visual estimation and laboratory determination of blood loss during the third stage of labour. Aust N J Obstet Gynaecol 1996 may; 36(2):152-4.
- Gross JB. Estimating Allowable Blood Loss: Corrected for Dilution. Anesthesiology 1983; 58:277-80.
- Bourke DL, Smith TC: Estimating allowable hemodilution. Anaesthesiology 1974; 41:609-12.
- Duthie SJ, Ven D, Yung GL, et al. Discrepency between laboratory determination and visual estimation of blood loss during normal delivery. Eur J Obstet Gynaecol Reprod Biol 1991; 38:119-24.
- Baronofsky ID, Treoar AE, Wangensteen OH. Blood loss in operations: a statistical comparison of losses as determined by the gravimetric and calorimetric methods. Surgery 1946; 20:761-9.
- Kaplan S. Method of measuring blood loss. Anaesthesia 1978; 33:191-2.
- Leveen HH, Rubricus JL. Continuous, automatic, electronic determinations of operative blood losss. Surg Gynecol Obstet 1958; 106:368-74.
- Hare GMT, Tsui AKY, McLaren AT, et al. Anaemia and cerebral outcomes: many questions, fewer answers. Anaesth Analg 2008; 107:1356-70.
- Hendrickson JE, Hillyer CD. Noninfectious serious hazards of transfusion. Anaesth Analg 2009; 108:759-69.
- Bower WF, Jin L, Underwood MJ et al. Peri-operative blood transfusion increases length of hospital stay and number of postoperative complication in non-cardiac surgical patients. Hong Kong Med J 2010 Apr; 16(2):116-20.
- Hill GE, Frawley WH, Griffith KE et al. Allogeneic blood transfusion increases the risk of postoperative bacterial infection: a meta-analysis. J Trauma 2003 May; 54(5):908-14.
- Lee MH, Ingvertsen BT, Kirpensteinjn J et al. Quantification of surgical blood loss. Vet Surg 2006; 35(4): 388-93.
- Brecher ME, Monk T, Goodnough LT. A standardized method for calculating blood loss. Transfusion 1997; 37:1070-4.
- Razvi K, Chua S, Arulkumaran S et al. a comparison between visual estimation and laboratory determination of blood loss during the third stage of labour. Aust N Z J Obstet Gynaecol 1996 May; 36(2): 152-4
- Patel A, Goudar SS, Geller SE et al. Drape estimation versus visual assessment for estimating postpartum hemorrhage. Int J Gynaecol Obstet. 2006 Jun; 93(3) 220-4.
- Rose P, Regan F, Paterson-Brown S. Improving the accuracy of estimated blood loss at obstetric haemorrhage using clinical reconstructions. BJOG. 2006 Aug; 113(8): 919-24.
- Meiser A, Casagranda O, Skipka G et al. Quantification of blood loss. How precise is visual estimation and what does its accuracy depend on? Anaesthetist. 2001 Jan; 50(1): 13-20.
- Suresh J, Duthie, Dip. Ven et al. Discrepency between laboratory determination and visual estimation of blood loss during normal delivery. European Journal of Obstetric & Gynecology and Reproductive Biology 1990; 38:119-24.
First Published September 2011
Copyright Priory Lodge Education Limited 2011-
Click
on these links to visit our Journals:
Psychiatry
On-Line
Dentistry On-Line | Vet
On-Line | Chest Medicine
On-Line
GP
On-Line | Pharmacy
On-Line | Anaesthesia
On-Line | Medicine
On-Line
Family Medical
Practice On-Line
Home • Journals • Search • Rules for Authors • Submit a Paper • Sponsor us
All pages in this site copyright ©Priory Lodge Education Ltd 1994-


