Browse through our Journals...
TRYPANOSOMOSIS DUE TO TRYPANOSOMA EVANSI IN GAZELLES
Walter Tarello, DVM
Al Wasl Veterinary Clinic
P.O. Box 75565
DUBAI (United Arab Emirates)
This work has been done in the International Veterinary Hospital of Ahmadi, Kuwait
SUMMARY
Trypanosoma evansi infection was diagnosed in a herd of Arabian dorcas gazelles (Gazella dorcas saudiya) and in one Sand gazelle (Gazella subgutturosa manica) from Kuwait, showing some clinical signs of animal trypanosomosis, such as sudden death and paresis of hindquarters. Following a treatment with melarsomine the clinical signs disappeared in the Sand gazelle and the periodic occurrence of sudden deaths stopped in the herd of Dorcas gazelles. On our knowledge this is the first report on Trypanosoma evansi infection in gazelles with a description of symptoms and of an effective therapy.
Key-words
Trypanosomosis, Trypanosoma evansi, gazelle, Kuwait, melarsomine.
INTRODUCTION
Trypanosoma evansi is a protozoal parasite of mammalian blood mechanically transmitted by biting flies of the genera Tabanus, Lyperosia, Stomoxys and Atylotus (Brun and others 1998). Up to now, this is the only trypanosome parasite recorded in camels, dogs (Al-Taqi 1989) and cats (Tarello 2005) from Kuwait. T. evansi is pathogenic in most domesticated animals and in some wild animals, including deer, elephants, capybara and jaguars (Brun and others 1998). However, naturally occurring trypanosomosis has never been recorded in gazelles. The aim of this study is to describe the associated clinical signs and the response to melarsomine (Cymelarsan, Merial) in a herd of captive Arabian Dorcas gazelles (Gazella dorcas saudiya) and in one Sand (‘Reem’) gazelle (Gazella subgutturosa manica) diagnosed with Trypanosoma evansi in Kuwait.
CASE HISTORY 1
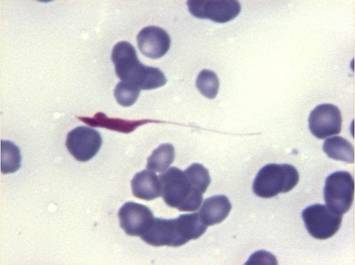
During August 2003, eleven Arabian Dorcas gazelles living in a horse-breeding farm in Wafra (Kuwait) were visited and tested for T. evansi infection. This group was the remnant of 20 animals, 9 of which died suddenly during the last 6 months without any clinical explanation or premonitory signs. The first gazelle, a 2-year old male, was visited on August 18 because of a 1-day history of anorexia, lethargy, and collapse. Rectal temperature was normal. A diagnosis of T. evansi infection was based on detection of trypomastigotes in fresh blood smears stained with the Wright technique (Fig.1).
Fig. 1) Trypanosoma evansi trypomastigote in the blood of a Dorcas gazelle (x100, Wright stain).
A cat living in the same farm was previously diagnosed with trypanosomosis (Tarello 2005). Treatment with melarsomine (Cymelarsan, Merial) was started immediately, but the gazelle died one hour later (Fig 2). The second gazelle, a 1-year old female, was examined soon after death on August 24.
Fig. 2) A Dorcas gazelle (Gazella dorcas saudiya) with trypanosomosis few minutes before death.
Blood smears obtained by heart-puncture revealed the presence of a high number of trypomastigotes. Five apparently healthy gazelles from the same herd were then captured and brought for examination on August 26. Three females resulted negative and two males positive for T. evansi. Shapes and lengths of the parasites were highly pleomorphic. All five animals were subsequently treated with a single injection of melarsomine at the recommended dosage of 0.25 mg/Kg of body weight (Zablotskij and others 2003). A last group of four apparently healthy gazelles was examined on August 28 and all were found negative for T. evansi. Nevertheless, the animals were treated, on a preventive basis (Seidl and others 2001), with a single injection of melarsomine. Gazelles were not re-tested because of the difficulty of recapturing them. However, no more deaths occurred in the herd during the following 7 months.
CASE HISTORY 2
In May 2004, a 2-year old female Sand (‘Reem’) gazelle (Gazella subgutturosa marica) was presented with a 3-weeks history of progressive paresis of hindquarters, showing difficulty in standing and walking.
The gazelle lived in a fenced garden of Kuwait City together with 11 gazelles of the same species. The animal had been unsuccessfully treated with antibiotics during the last 10 days. A Wright-stained blood smear revealed the presence of highly pleomorphic circulating trypomastigotes (Fig. 3 and 4) and a diagnosis of T. evansi infection was then made, followed by treatment with melarsomine at the same dosage used in the previous cases.
Fig. 3) Trypomastigote in the blood of a Sand (‘Reem’) gazelle (x100).
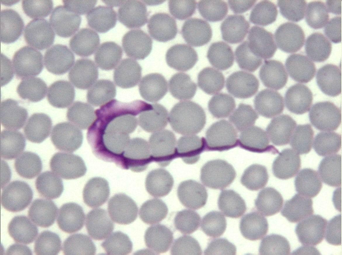
Fig. 4) Trypomastigote in the Sand (‘Reem’) gazelle (x100, Wright stain)
After therapy, the animal progressively improved his ability to stand and to walk (Fig. 5). At day 6 a blood check for trypomastigotes proved negative and the animal was able to stand and to walk without difficulties.
Fig. 5) Sand gazelle Reem with trypanosomiasis unable to stand and walk.

DISCUSSION
It has been noted that gazelle serum allows Trypanosoma brucei replication (Black and others 1999). However, to this author’s knowledge, there is no mention in literature of natural infection with T. evansi in gazelles. This is apparently the first report on this topic.
It might be argued that the here reported evidence for T. evansi as a cause of sudden death is circumstantial. However, the parasite is recognized as pathogenic in many wild animal species and can be acutely fatal in some cases (Brun and others 1998). In case history 1, two sudden deaths were directly linked to the finding of T. evansi trypomastigotes in the blood as sole detectable anomaly.
Nine unexplained deaths recorded during the 6 months before examination and the lack of mortality recorded during the 7 months following trypanocidal treatment may also account for such a link. Taken together, these facts strongly suggest that T. evansi was implicated in the occurrence of sudden deaths in the herd.
In case history 2, the link between the detection of trypomastigotes and clinical signs is more evident due to the rapid clinical improvement observed after melarsomine administration and also because progressive paresis of hindquarters (Fig. 5) is a common sign of trypanosomosis in many species (Brun and others 1998). In fact, alterations in muscle fibres and capillary necrosis were observed in infections due to T. evansi in wild horses (Quinones Mateus and others 1994) and might also account for heart infarction causing sudden death in gazelles. T. evansi is difficult to distinguish morphologically from other members of the subgenus Trypanozoon, such as Trypanosoma equiperdum (Zablotskij and others 2003) and Trypanosoma brucei (Masiga and Gibson 1990).
However, up to now T. evansi is the sole trypanosome isolated from camels, dogs (Al Taqi 1989) and cats (Tarello 2005) in Kuwait. Identification of the parasite by blood smear is recommended by the OIE (OIE 2004). Serology alone may not distinguish between current and past infections.
For these reasons, diagnosis in these cases was based uniquely on the microscopic detection of trypomastigotes and the response to specific medication. Where distribution areas of animal trypanosomes overlap it would be preferably to confirm the diagnosis using serological tests such as enzyme-linked immuno-absorbent assay (ELISA) and the indirect fluorescent antibody test (IFAT) (Brun and others 1998).
In the Equidae, the incubation period varies from 5 to 60 days and the disease is often fatal within two weeks to four months, with symptoms such as anaemia, intermittent fever, weakness, petechial haemorrhages, oedematous swelling of the legs and abdomen, urticaria, progressive paresis of hindquarters and death (Brun and others 1998). No search for trypomastigotes was performed on thirty horses living on the farm where case history 1 originated. However, one seriously ill cat living in the same area was previously diagnosed with trypanosomosis (Tarello 2005) confirming the existence locally of a disease reservoir. Furthermore, during the following 7 months, no unusual clinical signs or sudden death were reported in the herd.
In summary, this study seems to indicate that trypanosomosis may occur in gazelles. Clinical signs including progressive paresis of hindquarters and sudden death were recorded and melarsomine proved to be useful in its treatment.
REFERENCES
Al TAQI M.M. (1989) Characterization of Trypanosoma (Trypanozoon) evansi from dromedary camels in Kuwait by isoenzyme electrophoresis. Veterinary Parasitology, 32, 247-53.
BLACK S.J., WANG Q., MAKADZANGE T., LI Y.L., VAN PRAAGH A., LOOMIS M. & SEED J.R. (1999) Anti-Trypanosoma brucei activity of non-primate zoo sera. Journal of Parasitology 85, 48-53.
BRUN R., HECKER H. & LUN Z.R. (1998) Trypanosoma evansi and T. equiperdum: distribution, biology, treatment and phylogenetic relationship (a review). Veterinary Parasitology 79, 95-107.
MASIGA D.K. & GIBSON W.C. (1990) Specific probes for Trypanosoma (Trypanozoon) evansi based on kinetoplast DNA minicircles. Molecular and Biochemical Parasitology 40, 279-83.
OIE. (2004) Trypanosomosis. In: Manual of standard for diagnostic tests and vaccines 2004. www.oie.int/eng/normes/mmanual/a_00093.htm Accessed 28 January 2007.
QUINONES MATEUS M.E., FINOL H.J., SUCRE L.E. & TORRES S.H. (1994) Muscular changes in Venezuelan wild horses naturally infected with Trypanosoma evansi. Journal of Comparative Pathology 110, 213.
SEIDL A.F., MORAES A. S. & SILVA R.A. (2001) Trypanosoma evansi control and horse mortality in the Brazilian Pantanal. Memorias do Instituto Oswaldo Cruz 96, 599-602.
TARELLO W. 2005) Trypanosoma evansi infection in three cats. Revue de Medecine Veterinaire 156, 133-134.
ZABLOTSKIJ V.T., GEORGIU C., DE WAAL T., CLAUSEN P.H., CLARES F. & TOURATIER L. (2003) The current challenges of dourine: difficulties in differentiating Trypanosoma equiperdum within the subgenus Trypanozoon. Revue Scientifique et Technique 22, 1087-96.
Click
on these links to visit our Journals:
Psychiatry
On-Line
Dentistry On-Line | Vet
On-Line | Chest Medicine
On-Line
GP
On-Line | Pharmacy
On-Line | Anaesthesia
On-Line | Medicine
On-Line
Family Medical
Practice On-Line
Home • Journals • Search • Rules for Authors • Submit a Paper • Sponsor us
All pages in this site copyright ©Priory Lodge Education Ltd 1994-


