‘Acute respiratory failure’ in a previously healthy young female
Chaminda Jayawarna,
Medical Registrar, Tameside General Hospital, Ashton-under-Lyne. UK
Syed V Ahmed
Consultant Physician, Tameside General Hospital, Ashton-under-Lyne. UK
Abbreviations: HRCT- high resolution computerised tomography, ANCA- anti-neutrophil cytoplasmic antibody, ACE-angiotensin- converting enzyme.
Keywords: Lymphangitis carcinomatosis, respiratory failure, lymphatic infiltration, gastric malignancy, hypoxia.
A previously fit and well 46 year old lady presented with 3 weeks history of progressively worsening dyspnoea and a dry cough. On admission she was orthopnoic and breathless at rest, barely able to walk a few steps. She did not have haemoptysis or chest pain. Her appetite was normal with no recent weight loss. There were no rashes or any joint involvement. Before admission, she had a course of antibiotics and steroids with no effect.
Apart from lumpectomy for a benign breast lesion, her past medical history was unremarkable. She never smoked and there was no exposure to birds or pets. She had not been on any regular medications. There was no significant family history or a contact history of a similar illness and no recent travel abroad.
On examination, she was afebrile and tachypnoeic with a respiratory rate of 32 per minute. She was not cyanosed and there were no clubbing, rashes, palpable lymph nodes or dependent oedema. Pulse rate was 132 beats per minute and blood pressure was normal. Jugular venous pressure was not raised and cardiovascular examination was normal. She had oxygen saturation of 88% on air while arterial blood gas on 35% oxygen by facemask showed pH 7.48, pCO2 5.5, pO2 8.1 with base excess -6.4. Chest examination revealed bilateral widespread fine crackles. Liver was not palpable and the rest of the examination was normal.
Her laboratory investigations showed haemoglobin13g/dl, white cell count16.8x109/l, neutrophils14.5x109 /l with normal eosinophils, lymphocytes and platelets. Urea, creatinine, electrolytes were normal while corrected calcium was 2.83mmol/l. Blood cultures were sterile though C-reactive protein was elevated to 96mg/l. Her liver function tests were normal with total protein 51.5g/l and albumin decreased to 18.9g/l. Connective tissue disorder screen was negative, including ANCA, antiGBM and rheumatoid factor with normal C3/C4 compliment levels. Antibodies to Cytomegalovirus, Epstein-Barr and Toxoplasma were not detected. The serum ACE levels were normal.
Her CXR (Fig 1.1) revealed diffuse bilateral reticular infiltrates and prominent septal lines compatible with interstitial oedema. An X-ray done 2 weeks earlier was normal except a few linear opacifications in the right mid zone and both bases. Her pulmonary function tests showed severe restrictive type of disease.
On echocardiogram, she had normal left ventricular systolic function with moderate enlargement of right heart chambers and moderate tricuspid regurgitation. Pulmonary artery pressure was 47 mmHg + Jugular Venous Pressure.
High-resolution computed tomography scan (HRCT) of the Thorax showed areas of groundglass opacifications in both lung fields with some patchy confluent bilateral opacifications in the upper lobes and to a lesser extent in lower lobes. Pleural thickening was noted in the left lower zone, subjacent to an area of parenchymal opacification. Peripheral interstitial thickening was noted bilaterally, most likely representing septal lines. There was no mediastinal lymphadenopathy.
(Fig 1.2)
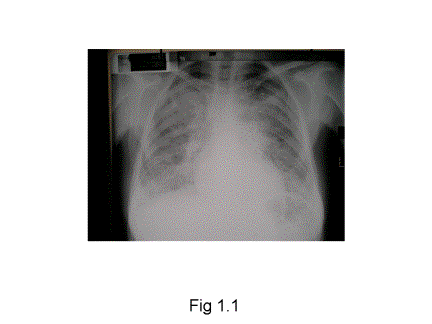
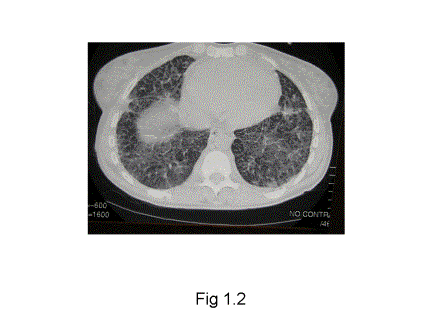
Our initial diagnosis was interstitial lung disease with secondary pulmonary hypertension and superadded infection resulting in respiratory failure. She was treated with antibiotics and high dose steroids pending results of specific investigations. Unfortunately, she continued to deteriorate rapidly and died 3 days following admission.
At autopsy, both lungs appeared heavy and cut surfaces were solid and consolidated. There was a solid congested, slightly mucoid nodule of 4 cm in diameter in the upper lobe. Examination of the stomach showed a large ulcerated antral tumour with full thickness invasion and spread to the lymph nodes along the lesser curvature. Liver did not show any metastatic involvement.
Microscopic sections revealed a locally arising poorly differentiated adenocarcinoma extensively infiltrating the entire thickness of the stomach wall. (Fig 2) Sections from both lungs showed large deposits of metastatic adenocarcinoma with extensive embolisation of lymphovascular spaces compatible with lymphangitis carcinomatosis. The surrounding parenchyma showed organising pneumonia.

Discussion:
Lymphangitis carcinomatosis represents 7% of all pulmonary metastases. However, in postmortem studies, the incidence is much higher with microscopic
tumor invasion as high as 56%.
It is a diffuse metastatic infiltration and obstruction of pulmonary lymphatics by a tumour residing elsewhere, such as breast, lungs, pancreas, prostate, colon, and gastric malignancies1. About 80% of this type of metastatic disease is from adenocarcinomas. It is generally believed that such infiltration occurs as a result of lymphatic spread to the lungs due to tumour microembolization1 which ultimately leads to pulmonary hypertension and the development of cor pulmonale2.The exact incidence of lymphangitic spread is not known since most cases are discovered at autopsy2. It carries a very poor prognosis.
Typically, the patient has been treated for a primary malignancy by the time the lymphangitis carcinomatosis is diagnosed. Occasionally, it may be the presenting feature with an occult malignancy. The most striking presenting feature is progressive, unrelenting dyspnoea1 with or without significant cough and haemoptysis. Symptoms often precede any radiological abnormality. Physical examination reveals tachypnoea, tachycardia and diffuse crackles on chest examination without other evidence of consolidation 5.
Plain radiography usually shows a coarse reticulonodular pattern, more obvious in the lower zones. However, the abnormalities are mostly disproportionate to the symptoms, and CXR may even be normal6. Occasionally, Kerley B lines are seen without any features of congestive cardiac failure5. High Resolution CT thorax (HRCT) is useful, even though it is not diagnostic. HRCT findings include circumferentially thickened and irregular or nodular bronchovascular bundles, multiple linear densities forming a reticular network, and the presence of well defined polygonal lines6. The differential diagnoses for this type of radiographic findings include extrinsic allergic alveolitis, cryptogenic fibrosing alveolitis, lymphoma, Kaposi’s sarcoma and sometimes sarcoidosis. In some patients, transbronchial biopsy is the diagnostic procedure of choice7.
Hypoxia is a very common finding resulting either due to tumour emboli8 or compression of the adjacent vessels by the distended lymphatics3. HRCT is particularly useful in differentiating it from pulmonary emboli due to venous thromboembolism.
Mean survival after presentation is only 2 months9. Currently, data regarding management of lymphangitis carcinomatiosis are lacking and there are no definitive treatment options available. The value of chemotherapy has been disputed though a few case reports suggested promising results and extended life expectancy4, 10. Currently, only palliative care is recommended.
Our case was extremely unusual; firstly, it was an acute presentation with a relentless course and death in less than 4 weeks with no previous symptoms suggestive of primary malignancy, particularly in association with undiagnosed carcinoma of stomach. Secondly, the autopsy confirming dense bilateral consolidations as a manifestation of lymphangitis carcinomatosis has been described once previously3.
Learning points:
- Lymphangitis carcinomatosis can present as ‘acute respiratory failure’ as a first manifestation of underlying malignancy.
- Needs high index of suspicion in cases with unexplained relentless dyspnoea with past history of malignancy.
- Even if diagnosed, the most challenging part is to treat it, as no definitive treatment options are available other than managing symptoms.
- Therefore, it is a matter of urgency to look for some form of treatment that could prolong, if not cure, the current hopeless mean survival of 2 months after diagnosis.
Acknowledgments: Dr Neha Dalal, Consultant Clinical Pathologist, Tameside General Hospital, Ashton-under-Lyne.
References:
- Maza I, Braun E, Plotkin A, et al. Lymphangitis Carcinomatosis of Unknown Origin presenting as severe pulmonary hypertension. American Journal of the Medical Sciences 2004; 327(5): 255-257
- Molina D K, Valente P T. Lymphangitic spread of Hepatocellular Carcinoma. Arch Pathol Lab Med. 2003; 127:e11-e13
- Sood N, Bandarenko N, Paradowski L J. Acute respiratory failure secondary to Lymphangitis Carcinomatosis. Journal of Clinical Oncology 2000; 18(1): 229 – 232
- Fujita J, Yamagishi Y, Kubo A, et al. Respiratory failure due to pulmonary lymphangitis carcinomatosis. Chest 1993; 103: 967-8
- Fraser RG, Pare J A P, Pare P D, et al: Diagnosis of Diseases of the Chest (edition 3). Philadelphia P A, Saunders W B, 1991
- Munk PL, Muller NL, Miller R, et al: Pulmonary lymphatic carcinomatosis CT and pathologic findings. Radiology 1988; 166 : 705-709
- Arando C, Sidhu G, Sasso LA, et al: Transbronchial lung biopsy in the diagnosis of lymphangiatic carcinamatosis. Cancer 42: 1978; 1995-1998
- Gonzales-Vitale JC, Garcia-Bunuel R: Pulmonary tumour emboli and cor pulmonale in primary carcinoma of the lung. Cancer 38:1976; 2105-2110
- Hauser T E, Steer A. Lymphangitis carcinomatosis of the lungs: six case reports and a review of literature. Ann Intern Medicine 1951; 34:881
- Ikezoe J, Godwin J D, Hunt K J, Marglin S I. Pulmonary lymphangitic carcinomatosis: chronicity of radiographic findings in long-term survivors. Am J Roentgenol 1995;165 : 49-52
Home • Journals • Search • Rules for Authors • Submit a Paper • Sponsor us
All pages copyright ©Priory Lodge Education Ltd 1994-

