James V. Schoster, DVM DACVO
Fox Valley Animal Referral Center
842 Westhill Boulevard
Appleton, WI 54914 USA
jschoster@horizondvm.com
CHRONIC SUPERFICIAL CORNEAL EROSIONS
IN ANIMALS
![]()
James V. Schoster, DVM DACVO
Fox Valley Animal Referral Center
842
Westhill Boulevard
Appleton, WI 54914 USA
jschoster@horizondvm.com
INTRODUCTION
Chronic non-healing superficial corneal erosions can be frustrating to the client, patient and veterinarian. These special non-healing lesions may or may not have been initiated by trauma. In many cases it appears that they have arisen spontaneously.
This syndrome can be characterized by the following:
1. A corneal epithelial erosion that tends to remain superficial.
2. The presence of a redundant or loose epithelial margin or bleb.
3. Fluorescein stain will undermine the redundant border or pass through a fine epithelial break and be retained under the epithelium beyond the edge of the surface break.
4. The loose epithelial margin may even be rolled or folded upon itself.
5. The eye may not be consistently painful and it usually is intermittently painful.
6. Initially and even chronically there is usually a lack of an inflammatory response characterized by a lack or scarcity of blood vessels.
7. These lesions rarely become infected.
8. They are commonly located in the central or paracentral cornea, yet they can potentially be found anywhere on the corneal surface.
9. The condition can occur in any species of animal, including man. Veterinarians are most often presented with dogs; especially Boxers and middle to older age dogs of any breed or sex, neutered or intact. The syndrome has also been seen in the cat and the aged horse.
Names that have been given to these lesions and this condition are:
rodent ulcers, Boxer ulcers, indolent ulcers, refractory ulcers, non-healing corneal ulcers, persistent corneal erosions, recurrent erosions, recurrent epithelial erosions, recurrent ulcers and many others.
The etiology is not precisely known in the majority of the cases. Over the past ten years much work has been done to investigate this problem. Out of this work a greater understanding about its nature is emerging. References to this syndrome in humans goes back as far as 100 years and thirty years in the Boxer dog.
At this time it does seem clear that there is a fundamental lack of adherence of the corneal epithelium to the anterior corneal stroma. After a corneal epithelial break, no matter how proliferative the new epithelium is, there is a failure of the new epithelium to securely fuse with the anterior stroma which allows the shearing force of the eyelid action to elevate this loosely bound epithelium and fold or roll it back upon itself.
In humans, physicians have identified several etiologies. Two common ones are trauma from a sharp superficial cut (paper or fingernail) that can lacerate the epithelium and excise a piece of basement membrane. Some patients then do not replace this lost piece of basement membrane and their lesion becomes indolent.
The second most common etiology is a basement membrane dystrophy of known or unknown etiology or secondary to aging where the basement membrane duplicates with collagen packets in between resulting in abnormal epithelial adhesions.
Some other etiologies in man include microcystic epithelial degeneration complicated by hypotonic tears from closed eyelids during sleep. Diabetes mellitus with basement membrane abnormalities and many other corneal dystrophies that affect the basement membrane have been implicated.
In animals, we recognize that the animals with corneal dystrophy are at greater risk for indolent ulcers. Yet, clinically we see many animals with indolent ulcers that do not have any gross corneal changes that would imply there is a dystrophy. Because of the greater incidence of this problem in older animals an age related basement membrane abnormality may be playing a role. The Boxer definitely has a greater incidence than in any other breed and a microscopic basement membrane dystrophy is involved in these animals.
NORMAL ANATOMY AND PHYSIOLOGY
Focusing in at the junction of the anterior stroma with the corneal epithelium, the literature describes an incredibly complex physical chemical bond.
There are not only physical bonds such as hemidesmosomes that bridge the basement membrane and anchor at either end (basal epithelium and anterior stroma) but there are chemical bonds composed of complex macromolecule glycoproteins called fibronectin and laminin.
The physiology of this union is very complex and during normal healing of an epithelial defect these bonds are broken down to allow for epithelial sliding. Normally, once the defect is covered, new firm bonds occur. It has been theorized that in the animals with persistent epithelial defects, this final firm bond does not develop and the non-union persists, irrespective of the proliferative capability of the epithelium.
Certain chemicals such as tissue plasminogen-activator (PA) that normally emerge to temporarily break these bonds to allow for epithelial sliding, may actually persist.
Animals with recognizable subepithelial anterior stromal dystrophies may have a predisposition to these persistent erosions, perhaps just due to the anterior stromal and/or basement membrane pathology.
PATHOGENESIS
Initiating factors, such as foreign bodies, trauma, aberrant lashes and cilia, eyelid neoplasms, tear film abnormalities, etc. can cause the original epithelial break. Many animals historically do not appear to have experienced any of the latter initiating factors and perhaps, in these, spontaneous tears may have developed due to the pre-existing poor epithelial bond, or possibly a wrinkle or blister of epithelium that develops, which is then torn open by normal eyelid movement over the corneal surface.
Once there is an epithelial defect and the epithelial margins fail to firmly adhere, eyelid movement over the cornea continuously tears back the epithelium in spite of the epithelium's continued proliferation and sliding to cover the wound.
CLINICAL COURSE
The during the clinical course of this syndrome there can be periods of relative ocular comfort when the epithelium has nearly covered the defect, yet is still loose. Clinical signs at this time may consist only of mild ptosis and epiphora. Abrupt acute pain usually results from the shearing forces of the eyelid movement over the unattached epithelium resulting in the lifting and tearing and/or rolling back of the loose epithelial margins.
This cycle may wax and wane for weeks, and even months, without much, if any, corneal neovascularization or inflammation. Medical therapy with antibiotics, sodium chloride ophthalmic ointments, artificial tears, contact lenses, third eyelid flaps, systemic androgens/estrogens, tetracycline, etc. seems to make no difference. Some will just spontaneously heal on their own, yet most will remain chronic.
DIAGNOSIS:
A cardinal sign is an erosion with a loose redundant epithelial margin and lack of a vascular response. Sodium fluorescein applied to the lesion will migrate under the overhanging border of epithelium and stain the anterior stroma up to the point of firm epithelial attachment. Nonrecurrent erosions will stain only up to the epithelial edge and the stain will not go under the epithelial margin.
DIAGNOSTIC CONSIDERATIONS AT THE FIRST VISIT
Irrespective of the "cardinal" appearance and diagnosis at this point, one must be sure to evaluate the globe and adnexa for any abnormalities that could be causing the persistent epithelial defect (even Boxers), such as foreign bodies, aberrant cilia, etc. (as listed previously).
A swab of the conjunctival cul-de-sac should be taken for bacterial culture early in the examination, prior to the instillation of any drops, in those eyes that exhibit an abnormal discharge other than just a serous tear.
If a Schirmer Tear Test is done, it too should be done at this point prior to topical anesthesia and drops to avoid confusion of the results.
Evaluation of the anterior segment for signs of uveitis should also be done, noting in particular, the size of the pupil and if there is an aqueous flare.
Once the clinician has classified this corneal lesion as one with the potential of being indolent, and has identified any other complicating or predisposing factors; therapeutic modalities and anticipated clinical course can be discussed with the client. The latter being as important as the evaluation and accurate diagnosis.
The client needs to be informed of the potential for a chronic and possibly exacerbated course, with prospects of other modes of therapy being instituted during the course of therapy, if progress is not being made. The therapeutic plan should start conservatively.
THERAPEUTIC MODALITIES
The redundant epithelial border must be removed irrespective of the initial etiology or what other medical or surgical modality is chosen to follow! Included in this debridement should also be the floor of the erosion to include the removal of allegedly abnormal basement membrane and complexes associated with it.
In humans, the clinician tries to separate out the post-traumatic from basement membrane dystrophy cases by slit lamp examination. In my experience with animals, unless the dystrophy is conspicuous, the inciting etiology still remains vague.In either case however, the treatment is usually the same, but the prognosis (especially for the fellow eye) is different. Most physicians begin with patching after instillation of a topical antibiotic and cycloplegic agent. If not successful, a soft Therapeutic Bandage contact lens is placed (i.e: Bausch and Lomb PlanoT Soflens). Some cases may require tarsorrhaphy, if only on a temporary basis. Fibronectin appears to be useful in recalcitrant cases. If the epithelial edge of a defect is folded or rolled back, it is often debrided. In patients with recurrent erosions and an intact epithelium, micropuncture through the intact epithelium with a sterile bent 27 gauge needle into anterior stroma in multiple locations has been utilized.
Veterinarians have also tried this method, but in a different way. Veterinary reports of epithelial debridement followed by multiple anterior stromal punctures have been made with allegedly more rapid healing afterwards. My impression of the veterinary modification of this micropuncture technique is that it is still questionable and there is a risk of full thickness penetration of the cornea with the needle.
DEBRIDEMENT OF THE REDUNDANT EPITHELIAL MARGIN, ADJACENT UNSTABLE OR EASILY DEBRIDEABLE EPITHELIUM AND IDEALLY, ANOMALOUS BASEMENT MEMBRANE AND COMPLEXES IS THE CLASSICAL INITIAL THERAPEUTIC APPROACH IN VETERINARY MEDICINE
The idea is to remove all of the truncated anomalous complexes and aberrant basement membrane along with the associated loose epithelium back to where the epithelium is firmly adherent and assumed normal. This will allow for the new wave of epithelial migration to cover over a "clean" anterior stroma not cluttered with abnormal basement membrane and complexes, and hopefully subsequently lay down "normal" basement membrane and firm complexes.
All modes of therapy that follow debridement are just methods to make the environment favorable for adhesion of the new sheet of corneal epithelium.
PROTECTION
PAIN RELIEF
PREVENTION OF INFECTION
THERAPEUTIC DETAILS
CORNEAL EPITHELIUM DEBRIDEMENT
Debridement can be accomplished in a variety of ways. All of the following techniques are essentially variations of a superficial keratectomy. There is no one best technique, in general, yet there is a best technique for the practitioner, based on their experience with one, or several of these methods.
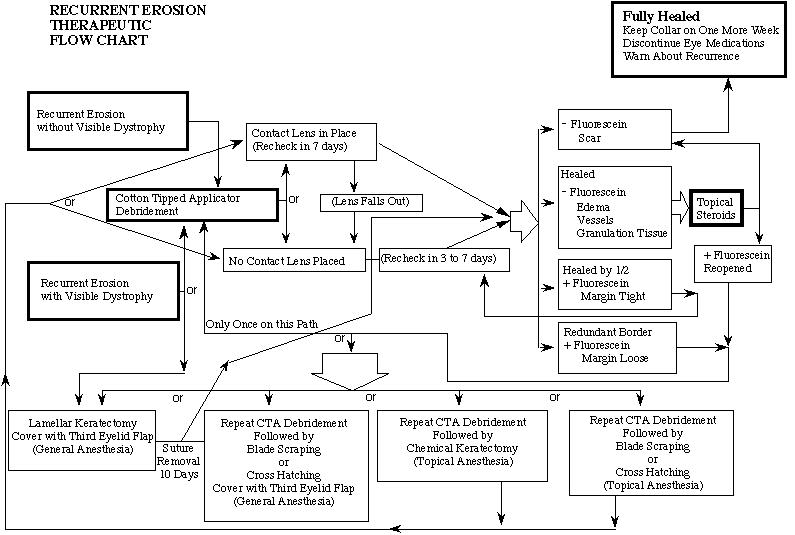
In other words, become acquainted with a method, rather than jump back and forth between methods. I prefer to start with one method on the first visit and only change methodology in that particular case to a more aggressive one if the cornea is not responding. Please see flow chart, Figure 1.
Examples of Debridement Methods That Have Been Described
Sterile Cotton Tipped Applicator (CAT's) debridement.
Scraping with a #15 Bard-Parker blade or #64 Beaver blade.
Superficial Lamellar Keratectomy with a #15 Bard-Parker blade or #64 Beaver blade.
Cross-hatching (scarification) with a 25 or 27 gauge hypodermic needle.
2% Tincture of Iodine
Trichloroacetic Acid (TCA)
Povidone Iodine Solution: half strength
For example, on the first visit the cornea is debrided with sterile cotton tipped applicators (CTA) followed by the application of a soft contact lens if available and if the fit is OK. If, during the course of subsequent re- evaluations the margins again become redundant, either a repeat of the CTA debridement and/or a scraping with a blade would be attempted in an animal that is cooperatable. If the animal is uncooperatable and a blade scraping is planned, a general anesthesia must be used. While the animal is under a general anesthetic, I always place a third eyelid flap after debridement.
One could instead substitute a chemical keratectomy for the blade scraping and eliminate the need for a general anesthetic.
If the cornea still does not respond after this, then general anesthesia, blade scraping, and third eyelid flap should be done.
Most of the indolent ulcers respond to the sterile cotton tipped applicator debridement. A soft contact lens greatly increases the chance of healing after the first debridement but it is not absolutely essential.
Corneal neovascularization during healing is not necessarily a good sign or desirable, as it would be with complicated corneal ulcers. The reason being, that along with these vessels there is epithelial edema, especially when there is a lot of granulation tissue as well.
The epithelial edema (as with microcystic edema in humans) predisposes or makes the epithelium more fragile and easy for the eyelid movement to cause "self debridement."
Therefore in the cases with neovascularization and/or edema, with or without granulation tissue, I find it beneficial to treat the animal with systemic steroids to reduce the keratitis. Do not use topical steroids. Dosing these patients at one-half milligram per pound divided twice daily for 5 days, then tapering the dose down over the next 5 days does not appear to complicate healing like topical steroids could. In fact, some of these vascularized, edematous lesions would not heal if the edema was allowed to persist.
Topical sodium chloride ophthalmic would seem to be beneficial, however, it is yet another drug that needs to be applied which will increase blepharospasm resulting in more frictional irritation to the cornea. 5% sodium chloride can be irritating and it does not treat the primary cause of the edema which is the keratitis. It certainly has a place in some cases where the epithelial edema is producing vesicles or bullae or the edema is not the result of inflammation, and therefore would be helpful.
COTTON TIPPED APPLICATOR DEBRIDEMENT METHOD
Irrespective of the method used, or state of the animal (topical anesthesia or general), the basic idea of debridement is the same. The initially visible loose epithelium is stripped back with dry sterile applicators starting at the central portion of the erosion and wiping toward the periphery. It is usually apparent that more than the initial amount of redundant epithelium strips off. That is OK. Firm, but gentle, pressure with the dry CTA's (exchanging for sterile dry ones as their tips get wet) is all that is usually necessary.
When the loose edge reaches a point where it is more firmly attached, rotate the tip of the swab that is holding the folded over piece of epithelium to coax it to tear off evenly.
Islands of firmly attached epithelium may remain and if they are firm and have tight clean margins, I generally leave them alone.
Too vigorous abrasion will remove even the normal epithelium. There is no need to remove the firmly adherent normal epithelium. It is also important to wipe dry applicators over the floor of the erosion to remove the abnormal basement membrane and its complexes. This might be where some of the other techniques are useful in the refractory cases. For example, at this point (after mechanical debridement) a chemical keratectomy would be done, if elected to do so, and the iodine or trichloroacetic acid may do a better job in removing these invisible membranes and complexes than frictional removal with a CTA. If I were to choose a chemical method to follow the mechanical, I would choose 2% tincture of iodine. Irrigation of the eye with eye wash must be done immediately after the cornea is wiped with the iodine.
The same mechanism of invisible abnormal membrane and complex removal likely is involved with the blade scraping or scarification with a needle (X hatching).
I reserve the chemical and blade debridements for the cases that do not heal with just the cotton tipped applicator debridement for two reasons.
Firstly, most erosions will heal after the cornea is properly debrided with cotton tipped applicators and therefore any added risk that is imposed with the other techniques seems unwarranted at the first visit. Secondly, the chemical keratectomy is irritating and evokes more of a reflex anterior uveitis which may not be necessary to subject the animal to on the first go around. Deep scarification (X-hatching) in a linear pattern has the potential of permanent scarring.
I would prefer to hold this technique in reserve until the standard conventional CTA debridement method has been done and failed.
Post Debridement Therapy
Protection (very important)
Soft Contact Lenses
After the cornea has been debrided the following regimen can be followed. If a soft contact lens is available, it should be used. Be sure reflex tearing is normal (the Schirmer values are greater than 15 mm/min.).
The contact lens will protect the cornea from the movements of the overlying eyelids and splint the new epithelium tight up against the anterior stroma. The fit of the lens needs to be carefully evaluated. Too loose of a fit (large base curve or radius of curvature) will result in lens loss due to wrinkles forming and the eyelid catching on it and lifting it out.
Too tight of a fit (small base curve or radius of curvature) can cause serious corneal damage due to blockage of the normal corneal respiratory cycle. Too small diameter of lens may result in lens loss from the margin of the third eyelid slipping underneath it and lifting it out. The lens should easily be seen to float freely in the tear film without any retained air bubbles or wrinkles.
Most dogs will comfortably wear a 14.5 mm to
15.0 mm diameter lens with a base curve of 8.6 to 8.8. Please see table 1 for a list of the available soft contact lenses that veterinarians have used successfully.
Any sign of increased ocular irritation, corneal cloudiness, etc. is of concern when a contact lens is in place and could mean that the lens is causing a problem or that there may be foreign
material trapped under the lens.
Third Eyelid Flap
Third eyelid flaps are an excellent form of protection and splint for the corneal epithelium during healing. Unfortunately, they almost always require a general anesthetic for placement. Therefore, I usually do not use them initially. If at any time a general anesthetic is required for debridement, I will then take advantage of the anesthetized situation and put a third eyelid flap up. If after the first debridement (or subsequent ones) healing does not occur and further more aggressive debridements are needed, I consider a third eyelid flap after the redundant epithelium is removed, unless a contact lens is available. If a contact lens fails (poor fit or lack of a healing response) I generally then consider a third eyelid flap after debridement. AVOID bilateral third eyelid flaps because, of course, the animal will then be acutely blind while the flaps are in place.
WYMAN MODIFIED THIRD EYELID FLAP
This modification involves suturing the third eyelid directly to the globe to prevent movement of the globe underneath and against the third eyelid which potentially could cause continued abrasion of the new corneal epithelium and result in non- healing.
Under general anesthesia, the third eyelid is grasped and extended in its natural prolapsed direction, toward the superior temporal fornix. The "T" portion of the cartilage is identified and a 4-0 braided nylon (Surgilene-D & G) is passed through the central "T" portion.
The bulbar conjunctiva near the fornix conjunctiva is grasped with a Bishop Harmon forceps or similar small rat-toothed instrument to a depth that feels firm (includes Tenon's and/or rectus muscle) and a wide .75-1 cm bite is deeply taken, avoiding penetration of the globe. The needle then passes out through the "T" portion of the cartilage about 4 mm away from the first bite. The ends are left long and not tied until all subsequent sutures are placed.
Two additional stitches are similarly placed in the wings of the "T", one on either side of the central stitch. Now they can be pulled up and tied. A surgeon's knot should be used. At least six tight throws must be placed on braided nylon, otherwise the knots will untie.
The third eyelid flap is left in place for about ten days. Most animals will tolerate suture removal with only topical anesthesia and physical restraint.
TOPICAL OPHTHALMIC MEDICATIONS
When a contact lens is in place
Topical ophthalmic solutions ONLY because ointments will clog pores of lens and reduce or eliminate the passage of respiratory gases, tear components, etc. through the lens.
Corneal edema will result when a lens blocks respiration and serious complications could occur rapidly.
Contact lenses will hold drugs against the cornea much longer than after just topical application and toxic reactions that ordinarily would not occur may be develop or be enhanced. These reactions can be due to the active ingredient and/or its preservative. Therefore, the frequency of application should be reduced to about half of what you would normally use for a particular drug if no lens were available.
Drugs
a. antibacterial: Either a triple antibiotic such as neomycin, bacitracin, polymyxin; gentamicin; or ciprofloxacin.
b. mydriatic-cycloplegic:Mydriatic-cycloplegic such as 1% atropine sulfate should only be used in the face of a uveitis. Occasionally, I will give one or two doses post- debridement because of the noticeable acute miosis (indicating an anterior uveitis [iritis and potentially cyclitis] secondary to the corneal debridement = axonal reflex involving the fifth cranial nerve and a release of agents by these corneal nerves that mediate release of prostaglandins). Uveitis is not usually a significant component in these cases and therefore it is not necessary to routinely use atropine.
c. Elizabethan Collar (use in every case).
Protects the eye from the animal rubbing at it with its paw. Needs to be used throughout and also for one week after the erosion has healed. This will not stop them from rubbing their head against the furniture.
When a contact lens is not in place.
Same as above, except ointments can be used.
The frequency is usually two to three times per day for the antibacterial. The frequency for the mydriatic-cycloplegic, is usually one to three times per day depending on the severity of the uveitis. The frequency for the sodium chloride would be three to four times per day.
In both of the above cases, the technique for application of the ophthalmic medications is important to avoid putting too much pressure against the globe and avoiding frictional irritation to the cornea by indiscriminate movement of the eyelids against the cornea.
DO NOT RUB IN THE MEDICATIONS
Owners always seem to want to rub in the eye medications after their application and this could be disastrous because of the frictional irritation from the movement of the eyelids over the delicate new epithelium.
The head should be stabilized and elevated by placing the muzzle in the area between the owner's thumb and index finger to act as a crutch while the other hand instills the medication from the medication container. The container should be held between the thumb and index finger while the outside edge of the same hand rests on the animals brow. The resulting three finger distance (middle through 5th digit) serves as a comfortable to hold the container tip from the eye during instillation.
The upper eyelid can then be elevated by pulling back on the brow skin with the lateral edge of the instilling hand while tilting the head up. At the same time the lower eyelid can be depressed with the thumb of the other hand, thus avoiding touching the eye and putting pressure against it.
Potential complications from improper medicating techniques include; contamination of the cornea and/or the medication bottle during administration, additional trauma to the cornea from the dropper tip, and painful finger pressure against the globe during treatment which also may also cause excessive eyelid pressure resulting in loss of the contact lens.
When ointments are used a similar instillation technique can be used. The ointment will exit the tube as a strip when the tube is cool and as a semiformed liquid when it is warm. Rate of exit from the tube can be disturbing to the clients and wasteful if the tube has been squeezed excessively prior to removing the cap and therefore handling these tubes as one would with a tube of toothpaste should be avoided. The tip of the tube is round and smooth but could still cause ocular trauma. Keeping the tube tip about a centimeter or two from the eye and allowing a strip or bleb of ointment to fall to the eye works well. Once the ointment strip touches the cornea, simply pull the tube quickly away from the eye. The surface adhesion strength of the ointment to the cornea is greater than the strength of the strip itself, and the ointment will break away from the tube quickly.
When a third eyelid flap is in place.
Ointments are preferred. They are carefully applied as a strip over the outer surface of the third eyelid.
There is no need to try to get the medication under the flap.
In most cases treatment with an antibiotic ointment at three times daily is sufficient.
FLUORESCEIN STAINING
a. Contact lens in place: fluorescein should not be applied with a contact is in place because the fluorescein will stain the lens. At the first (7-10 day) recheck after a lens has been placed, fluorescein does not need to be used, if:
1. The eye looks comfortable
2. The lens in place
3. The lens is clear and floating freely
4. The edge of the erosion may or may not be seen but does not look as if it is folded over upon itself
5. The lesion appears smaller.
Therefore in these cases I avoid disturbing the eye and just recheck them in a week.
If the edge of the lesion can not be seen and it appears to have healed, one can either remove the lens at this point or leave it in another 3-7 days and then remove it.
Do not leave a contact lens in place longer than 14 days!
(due to buildup of proteinaceous material in the lens pores leading to blockage of corneal respiration).
a. If at any time the eye seems more irritated and there is increased hyperemia and corneal edema, a cloudy contact lens, or the contact lens is not freely floating in the tear film; the lens should be removed and the cornea stained. Foreign material can get trapped under the lens and cause irritation.
b. If no contact lens was used; fluorescein should be applied...
VERY CAREFULLY
Wet the fluorescein strip with eye wash and allow a drop or two of the fluorescein solution to fall onto the eye.
DO NOT IRRIGATE THE EXCESS OUT!
Iatrogenic debridement of the new epithelium could occur at this point if the corneal is irrigated with eye wash. Normal tears will adequately remove excess fluorescein and it should be obvious whether there is retention or not.
Fully Epithelialized
Once the lesion has epithelialized it may take up to one month before the epithelium is firmly adherent. Some corneas will rapidly epithelialize and without any keratitis. In these cases, no further treatment is necessary other than advising the owner to keep the Elizabethan collar on for one additional week.
Epithelialized corneas (negative fluorescein) exhibiting a mild keratitis with edema, vascularization and granulation tissue will need a topical antibiotic steroid ophthalmic ointment starting at about three to four times per day and tapering the frequency over the next week or two until the keratitis resolves. Significant edema and granulation tissue may necessitate a topical sodium chloride ophthalmic ointment and or systemic steroids as well.
Rapid resolution of the nonulcerative keratitis is essential because the edematous epithelium is very prone to "self-debride" as a result of friction from the eyelids.
Prognosis
The prognosis is guarded in these cases because it is not uncommon for recurrences to occur any time in the future, in either eye. Especially in high risk animals such as Boxers and middle age or older animals or animals with a noticeable corneal dystrophy.
The response to therapy in each episode may not be the same. Just because the lesion was difficult to resolve the first time does not necessarily follow true with subsequent erosions and vice versa.
SUMMARY
This syndrome can be a pleasure to work with, once the clinician has an understanding of the problem, and its natural course or biologic behavior. This especially true when a complete ophthalmic exam has been performed, and a reasonable diagnostic and therapeutic regimen has been followed. Most importantly, counseling the client about the problem at the first visit and in doing so being careful not to make the future look dismal but optimistic because you can reassure them that you know what you are dealing with and what may be expected in the future.
Once a client is aware that these lesions can be problematic and the mode of therapy might change during the span of therapy, there will usually be no client communication problems if the animal requires repeated debridement and/or general anesthesia, surgical debridement and third eyelid flap, etc. Loss of these clients occurs when either the veterinarian has not accurately diagnosed the problem and treats the lesion as a simple traumatic erosion; which, if it does not heal as expected, these clients may seek the advise of another veterinarian. Therefore, be ready for animals presented that have been treated elsewhere and are not responding. These animals may have had debridement and still have not healed. There may be a more than usually intense keratitis as well. This inflammation may complicate healing until it can be reduced by means of systemic steroids (making sure that there are no other systemic problems that would contraindicate their use - i.e.: bacterial cystitis, etc.).
I have on occasion seen chronic cases that have been treated elsewhere for a significant period of time. When first presented to me the redundant corneal epithelium is extremely thick and leathery. There also may be a step from the limbus down to the redundant margin due to a "delta" of superficial vessels and granulation tissue. Even if this edge could be removed with a CTA, the step would complicate healing because of the physical drop down to the central cornea. These animals need to be anesthetized and a superficial lamellar keratectomy done to provide a more normal corneal profile. Some of these chronic cases may also develop a calcific plaque (band keratopathy or calcific corneal degeneration) that also requires a superficial lamellar keratectomy under general anesthesia. A modified third eyelid flap should be placed after the keratectomy. Certainly, if the cornea is less than one-half normal thickness after a keratectomy, a consideration of conjunctival flap should be made. These chronic keratitis corneas will also need systemic steroids to reduce the amount of keratitis and granulation tissue during the healing phase.
Some individuals have used a 360 degree conjunctival flap to cover the superficial corneal erosions after debridement, even early in the course of therapy. I personally feel that this is an over zealous plan, especially because the 360 degree flap is so traumatic. These flaps usually do not adhere to stroma because the new epithelium migrates in so fast.
However, there is a chance that a permanent adhesion could develop when the cornea is vascularized, which would permanently obstruct the visual path.
I reserve this technique for only the most refractory cases that have failed all other modes of therapy.
Dogs that have concurrent endothelial dysfunction can be the most challenging to manage; for, the corneal edema will compromise epithelial adherence due to subepithelial fluid accumulation. Topical hyperosmotic ophthalmic preparations [ointment when a contact lens is not in place and solution when there is a contact lens in place] (5% sodium chloride) are very helpful in these cases.
Aged animals may have endothelial cells that are marginally functional and any corneal inflammation may decompensate endothelial function which would be manifest as increased corneal edema. these cases not only benefit from topical hyperosmotic medications, but also systemic antiinflammatory drugs such as corticosteroids. Reduction in corneal edema will promote adherence of the epithelium. Unfortunately, animals with endothelial dysfunction not secondary to inflammation will not benefit from systemic corticosteroids.
FUTURE
In the future, drugs that will enhance or induce the new corneal epithelium to produce a stable adhesion will likely be available. Currently studies are being done with epidermal growth factor, fibronectin, plasmin inhibitors and other agents. Autologous serum used as a topical drop may be beneficial and has appeared to help in a few cases I have tried it in.
BIBLIOGRAPHY
1. Dice, P.F. and Cooley, P.L. Use of Contact Lenses to Treat Corneal Diseases in Small Animals. In Seminars in Veterinary Medicine and Surgery (Small Animal). Edited by Ketring, K.L. Grune and Stratton Harcourt Brace Jovanovich, Inc. Vol. III, No. 1, Feb. 1988:46-51.
2. Berman, M., Kenyon, K., Hayashi, K. and L'Hernault, N. The Pathogenesis of Epithelial Defects and Stromal Ulceration. In, The Cornea: Transactions of the World Congress on the Cornea III. Edited by Cavanaugh, H.D., Raven Press, New York. 1988:35-43.
3. Gelatt, K.N., Samuelson, . Recurrent Corneal Erosions and Epithelial Dystrophy in the Boxer Dog. Journal of the American Animal Hospital Association Volume 18 May/June 1982:454.
4. Kirschner, S., Niyo, D., Covitz, D., Betts, D. Ultrastructural Morphology of Persistent Corneal Erosions in Dogs: A Preliminary Report. In: The Proceedings of the American College of Veterinary Ophthalmologists, 1986.
5. Kirschner, S., Cooley, P., Kern, T., Munger, R., Nasisse, M. and Hartz. A. Epidermal Growth Factor for Treatment of Persistent Corneal Erosions. Abstract in, The Proceedings of the American College of Veterinary Ophthalmologists 1988:59.
6. Singh, G. and Foster, C.S. Treatment of Nonhealing Corneal Ulcers and Recurrent Corneal Erosions. In, The Cornea: Transactions of the World Congress on the Cornea III. Edited by Cavanaugh, H.D., Raven Press, New York. 1988:45-53.
7. Wood, T.O. and McLaughlin, B.J. Recurrent Erosion. In, International Ophthalmology Clinics. Volume 28, No. 1, Spring 1988:83-93.
8. Wyman, M. Ophthalmic Surgery for the Practitioner. In, Symposium on Surgical Techniques, Veterinary Clinics of North America: Small Animal Practice. Volume 9, No. 2. May 1979:333-340.