 |
 |
Most patients will have been smoking cigarettes for many years (probably in excess of 20 pack years). The two main symptoms of COPD are breathlessness and cough which may or may not be productive of purulent sputum. A history of persistent productive cough or recurrent infections especially in the winter months is common. The cough is usually worse in the mornings but bears no relationship to the severity of the disease. Excessive sputum volumes are unusual and may suggest bronchiectasis. Haemoptysis should alert the physician for the presence of a carcinoma of the bronchus as this is a frequent co-morbidity in patients with COPD, but is often just due to infective exacerbations
Breathlessness is a common feature of acute infective exacerbations, but breathlessness during normal every day activity develops insidiously over many years and most patients will have lost more than 50% of their predicted FEV1 by the time that breathlessness becomes a problem. Wheeze is often an accompanying feature of breathlessness and may be erroneously attributed to asthma.
Weight loss is common in patients with long standing disease with predominately emphysema (the old fashioned pink puffer), although weight gain may also be a feature suggesting chronic hypoxaemia and the onset of cor pulmonale (the blue bloater). Patients can rarely be classified as pink puffers or blue bloaters, and the two states do not have any pathophysiological correlation. Therefore the usefulness of this classification is questionable and its use is discouraged by both sets of guidelines.
There are no specific findings on examination, although signs of hyperinflation of the chest are highly suggestive of emphysema. These include a barrel shaped chest (increased antero-posterior diameter), use of accessory muscles of respiration, reduction of the cricosternal distance, tracheal tug, paradoxical indrawing of the lower ribs on inspiration (Hoover's sign), intercostal recession, hollowing out of the supraclavicular fossae, pursed lip breathing and reduced expansion. In addition the patient may have hyperresonant lung fields, prolongation of expiration, especially forced expiration >5s, and audible wheeze. None of these signs are specific to COPD and do not correlate very well with the severity of the disease which emphasises the need for objective assessment. Their presence, however, should alert the physician to the possible diagnosis of COPD.
As the disease progresses, signs of right ventricular dysfunction may develop (Cor pulmonale) because of the effects chronic hypoxaemia and hypercapnia which include peripheral oedema, raised jugular venous pressure, hepatic congestion, and the presence of metabolic flapping tremor. Despite the widely held belief that these signs are due to right ventricular failure, the pathophysiology cor pulmonale is likely to be due to altered renal function giving rise to salt and water retention rather than cardiac dysfunction secondary to pulmonary hypertension.
Both sets of guidelines emphasise the use of simple spirometric measurements to assess the severity and predict the prognosis of patients with COPD. However, they are less specific on the use of the other measurements of lung function in the diagnosis and management of COPD. It is probably good practice to perform routinely a minimum of spirometry with bronchodilator reversibility, static lung volumes, carbon monoxide gas transfer and pulse oximetry as baseline measurements. The addition of walk distance and a quality of life questionnaire may also be helpful as a baseline for future reference
In the European guidelines, the presence of mild airflow limitation is recognised by a reduction in the ratio of FEV1 to VC or FVC (<1.64 residual standard deviation below predicted FEV1/VC). In both sets of guidelines, severity is based on the measured FEV1 as a percent of predicted FEV1. The European guidelines define 3 stages of severity:
| 1. FEV1>=70% predicted : Mild |
| 2. FEV1=50-69% predicted : Moderate |
| 3. FEV1<50% predicted : Severe |
The American guidelines however stage the severity of COPD as:
| 1. FEV1>=50% predicted : Stage I |
| 2. FEV1=35-49% predicted : Stage II |
| 3. FEV1<35% predicted : Stage III |
Therefore a subject in the European Severe category may only be the equivalent of American stage II, and there is no European equivalent for the American stage III. This may lead to problems in the future when trying to compare trials in the managment of COPD patients. There is likely to be few patients recognised in the mild to moderate European stages since many of these patients will not be very symptomatic and will remain undiagnosed unless specifically screened for.
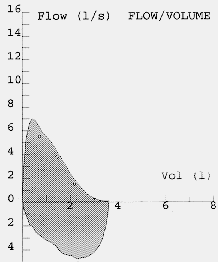
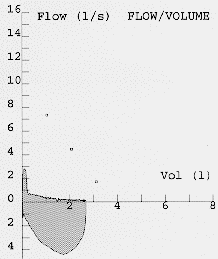
Most modern electronic spirometers will be able to produce an expiratory flow-volume curve or loop. The appearance of the flow volume curve is highly characteristic is airflow obstruction, especially when there is dynamic airways compression as in patients with predominately emphysema (fig 4). The flow volume loop in severe airflow obstruction is a good graphical representation of the severe impairment of airflow at low lung volumes due to dynamic airways compression. This information can also be obtained as absolute values from the spirometer. Neither set of guidelines emphasises the utility of flow volume loops as an aid to diagnosis although the appearances in severe disease are fairly specific. Severely obstructed flow volume loops may also be seen in obliterative bonchiolitis although there should be little difficulty in differentiating between the two on history alone.
 |
 |
Fig. 6: Flow volume loops showing normal and severe airflow obstruction
The absolute value of the FEV1 and the yearly rate of decline of the FEV1 may also provide information on the prognosis. The FEV1 at diagnosis can be a predictor of long term survival (fig 7). The yearly loss of FEV1 is about 20-30ml/yr after the age of 30 in normal non-smoking individuals. In smokers susceptible to developing COPD, the rate may increase to 50-90ml/yr (link to "Smoking and Bronchial Hyperreactivity) and fig 2) and thus this susceptible population can be identified and efforts increased to stop them smoking before developing disabling disease.
Fig. 7: Graph from Anthonisen NR et al, Am Rev Respir Dis 1986; 133:14-20
The use of peak expiratory flow rate (PEFR) for diagnosis is not recommended but may be useful for domiciliary monitoring or to document diurnal variation to differentiate from asthma. However, in severe COPD, the PEFR will tend to seriously underestimate the severity of airflow obstruction.
Most patients with COPD will show an increase in FEV1 in response to a bronchodilator, and some by more than 15% of the baseline value (which is often used as a diagnostic test for asthma), but never back to normal levels of lung function. In COPD. expressing reversibility as a percentage of baseline values is of limited value because of its dependence on the pretreatment level. Expression of reversibility as an absolute value or as a percentage of predicted values is more reproducible and independent of baseline FEV1. Improvement of the peak flow in response to bronchodilator is not recommended as a guide to reversibility. Many patients report subjective symptomatic improvement without any objective change in their spirometry. The absence of measurable spirometric reversibility therefore is not a reason to withhold bronchodilator treatment.
Some patients with stable COPD may show an increase in FEV1 following a prolonged trial of oral corticosteroids (e.g. Prednisolone 40mg od for 2 weeks). In Europe, this is often seen as an indication for continuing these drugs long term via the inhaled route. However, routine spirometry may not identify all responders to oral corticosteroids, since the improvement may be due to a reduction in FRC and an increase in FVC rather than FEV1. Therefore, there is case for measuring full lung function at the beginning and end of a trial of oral corticosteroids.
Total lung capacity (TLC), residual volume (RV) and functional residual capacity (FRC) are all characteristically increased in COPD and are related to the degree of hyperinflation of the lungs, especially when there is predominately emphysema. Although these measurements are useful and can help differentiate COPD from asthma, neither set of guidelines suggests that the use of these tests is essential in the diagnosis and managment of COPD. Similarly, although lung compliance is increased and lung recoil pressure is reduced and there are characteristic changes in the pressure-volume curve, these measurements are mainly research tools and not necessary in routine clinical practice.
Carbon monoxide gas transfer capacity (DLco)and coefficient (Kco) are both reduced in symptomatic patients with COPD. The transfer coefficient is a good indicator of the presence and severity of emphysema and thus reduction in the Kco is helpful in distinguishing patients with emphysema from those with asthma. There are also studies which relate the likelihood of hypoxaemia at rest and on exertion to the level of the Kco.
The relationship between symptoms, FEV1 and hypoxaemia is weak. The combination of FEV1 and gas transfer strengthens the prediction of resting hypoxaemia. Regular assessment of hypoxaemia is recommended in all patients with moderate to severe COPD (hypoxaemia is more likely when FEV1<1.0l). Rather than regular arterial blood gases analysis, it would be more sensible to use pulse oximetry as a screening test since this is a simple, cheap, painless and non-invasive technique which is fairly accurate. A reasonable strategy would be to perform pulse oximetry on all patients and perform arterial blood gas analysis only on patients with an arterial saturation of less than 93% (since this equates to PaO2 of about 8kPa or 60mmHg on the haemoglobin-oxygen saturation curve). Arterial blood gases should always be measured in patients with suspected CO2 retention. although this will rarely be present in the absence of arterial hypoxaemia and desaturation.
The presence emphysema can be suspected on routine chest radiography but this is not a sensitive technique for diagnosis. Large volume lungs with a narrow mediastinum and flat diaphragms are the typical appearances of emphysema Fig.8). In addition, the presence of bullae and irregular distribution of the lung vasculature may be present. In more advanced disease, the presence of pulmonary hypertension may be suspected by the prominence of hilar vasculature. The chest X-ray is not a very good indicator of the severity of disease and will not be able to identify patients with COPD without significant emphysema. However, the chest X-ray is useful to look for complications during acute exacerbations and to exclude other pathology such as lung cancer.
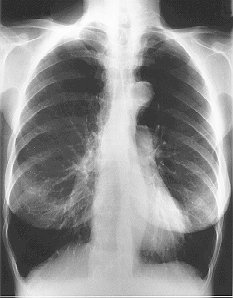
Fig. 8: Typical CXR of emphysema
Computerised tomography of the chest, especially with an high resolution algorithm (HRCT) has much greater sensitivity and specificity than plain chest radiography in diagnosing and assessing the severity of emphysema (fig 9). CT can identify areas of bullous disease that may be amenable to surgery that is not evident on plain chest radiography and is useful in predicting the outcome of surgery. HRCT is also capable of differentiating between the various pathological types of emphysema. However, the use of CT scanning in the routine clinical assessment of patients with COPD is not recommended by either set of guidelines, and is reserved for patients in which the diagnosis is in doubt, to look for co-existent pathologies and to assess the suitability of surgical intervention.
(Fig. 9: HRCT of emphysema)
The routine assessment of functional capacity is not recommended in either set of guidelines. This is rather surprising since often the main presenting symptom is limitation of walking and other activities of daily life by breathlessness. Since FEV1 and other measurements of lung function are poorly correlated to function capacity and subjective sensations of breathlessness, an objective measurement of the main presenting problem should be useful in helping to determine therapies that are actually beneficial. There may be little point in prescribing a treatment that may improve airflow obstruction, but the patient gain any improvement in functionality. Currently, the physician relies heavily on the the subjective sensations of the COPD patient to help determine the efficacy of therapy. Thus, there may be a case for the routine assessment of of functional capacity with simple tests such as six minute walk distance or shuttle walk distance in the managment of patients with moderate to severe COPD. The European guidelines states that the reproducibility of these tests is poor, yet they are frequently used as research tools in the development of new treatments for COPD. The use of more sophisticated tests of exercise performance such as VO2max should be limited to research or when the diagnosis is in doubt (i.e. when breathlessness is out of proportion to the degree of impairment of lung function).
There are several established questionnaires on quality of life (QoL) in chronic respiratory disease available (e.g. St.George's Hospital Respiratory Questionnaire, Chronic Respiratory Questionnaire) and other general health questionnaires with a respiratory component (e.g. SF-36, Nottingham Health Profile). Also, there are questionnaires on anxiety and depression which can contribute significantly to symptoms in COPD (Hospital Anxiety and Depression Score). These tools are frequently used in studies on the efficacy of new treatments in COPD but none have been accepted for use in everyday practice and have not been recommended for routine use. However, quality of life considerations are becoming increasingly important, and soon measures of QoL will soon be essential.
Many patients with COPD may have worsening hypoxaemia and hypercapnia during rapid eye movement (REM) sleep, and if this is combined with obstructive sleep apnoea it is called the Overlap syndrome. The role of nocturnal desaturation in the evolution of pulmonary hypertension is uncertain. Those who are most likely to desaturate at night are those who are already hypoxaemic during the day. Detailed sleep studies are currently only recommended in those with additional suspected obstructive sleep apnoea or those with cor pulmonale or polycythaemia with only mild or moderate COPD.